Infant mental health services for birth and foster families of maltreated pre-school children in foster care (BeST?): a cluster-randomized phase 3 clinical effectiveness trial
Study design and participants
We conducted a multi-site, pragmatic, parallel group, single-blind (researchers blinded to arm of trial), cluster-randomized controlled superiority trial with an allocation ratio of 1:1 to compare the NIM for infant mental health to social work SAU (NCT01485510) (see also the published protocol26). The trial was approved by West of Scotland Research Ethics Service, Committee 3 (15/WS/0280).
Inclusion criteria
Families were eligible for the trial if they had a child aged 0–60 months who entered care in the recruiting sites for reasons associated with maltreatment.
Exclusion criteria
Families were excluded from the trial if the parent(s) was unavailable to take part in an intervention (for example, because of death, unknown whereabouts or long-term imprisonment).
Study settings and context
The trial was conducted in two large UK cities, both containing areas of material deprivation. The study settings were social services in the local authorities/boroughs feeding into the two trial sites: Greater Glasgow and Clyde (Scotland), comprising Glasgow City Council and Renfrewshire Council, and London (England), including the boroughs of Croydon, Tower Hamlets, Sutton, Bromley and Barking & Dagenham. The social and legal contexts in the two sites varied considerably: in Scotland, legal decisions regarding children’s care placements are largely made by a Children’s Panel of lay members, although decisions about the presence/absence of maltreatment, and final adoption decisions, are made judicially by a Sheriff; in England, all legal decisions about children’s care placements are made within a court setting, presided over by a Judge11.
Randomization and masking
Random allocation was performed, through a web portal requiring a login and password with access rights allocated to relevant Clinical Trials Unit staff, using a mixed minimization/randomization method, stratified within the study site using an a priori schedule for each site in blocks of 10. Minimization factors were those likely to impact delivery of the NIM: study site, age of youngest child coming into care at randomization (<2/≥2 years), number of children coming into care at randomization (1/>1), birth family fluency in English and type of care (that is, foster or kinship (that is, with extended family)). Masking was assured for two secondary outcome measures, TTPLS and PIRGAS, because data were collected (TTPLS) and rated (PIRGAS) by individuals with no contact with participants or other trial procedures. Participants were aware of the intervention to which they had been allocated; data collectors, researchers and statisticians were not. Data collectors used a script, when conducting research assessments, aiming to ensure that participants did not reveal their group allocation, and, after assessment, data collectors recorded the intervention to which they thought the participating family was allocated. Trial group assignment was made available only when data collection was complete and after database lock.
Procedures
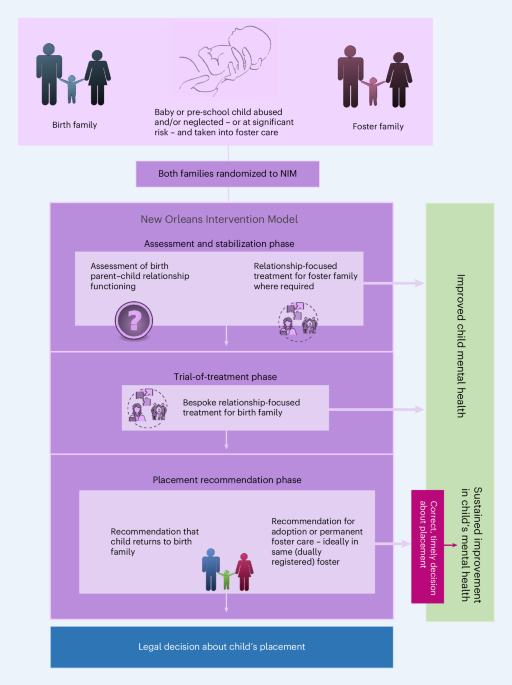
NIM (see panel) is an infant mental health intervention delivered by a multidisciplinary health and social care team. Families were eligible if they had a child aged 0–60 months entering care in the study sites for reasons associated with maltreatment. Children in eligible families entering care subsequently were allocated (rather than randomized) to the same intervention as the family had originally received. Families were excluded if parents could not receive interventions because of, for example, death or long-term imprisonment.
We used a novel recruitment and retention method: experienced social workers (former team leaders) screened social care records for eligibility and conducted information and consent meetings with interested participants. From January 2012 to June 2015, participants were asked for consent, assessed at baseline and then randomized. Thereafter, certain changes to randomization procedures were necessary (Supplementary Table 9): to give participants more time to consider participation, between June 2015 and September 2017 randomization was conducted first, and written deferred informed consent was obtained as soon as possible after randomization and before baseline assessment26 (Supplementary Table 9a). This caused further delays and problems with NIM capacity, and, when the English site joined the study, the only legally justifiable option was for families to be consented and randomized before baseline assessment11.
Outcomes
Study measures are detailed and referenced in Supplementary Table 10. Because NIM aims to improve the mental health of the infant or pre-school child by offering relationship-focused interventions making a recommendation to the legal system about the child’s permanent placement (Fig. 1), primary and secondary outcome measures focused on mental health/quality of life, relationship functioning and permanency. The primary outcome measure was the 20-item SDQ-TD scale at 2.5 years after study entry, assessed at each site. The SDQ is a mental health screening questionnaire for 2–16-year-old children, completed by the primary caregiver, with 25 items in five subscales: emotional symptoms, conduct problems, hyperactivity/inattention, peer relationship problems and prosocial behavior. The SDQ-TD does not include the prosocial subscale and contains 20 items. Higher SDQ-TD scores indicate poorer child behavior27.
Secondary outcome measures were as follows:
-
Child quality of life (PedsQL)28
-
TTPLS being achieved, authorizing the child to be placed in permanent care (return home without social work oversight or adoption)26
-
Quality of the child’s relationship with the primary caregiver (PIRGAS). For PIRGAS, a short playtime and mealtime video was conducted in the research clinic and independently rated by specifically trained raters29.
Study data were collected following standard operating procedures by trained staff. For interview and video measures requiring rating, more extensive training was provided until raters reached sufficient reliability, and then inter-rater reliability was checked on 10–20% of randomly selected tests, and, if insufficient agreement, ratings were brought to an approximately three-monthly conference with an expert rater (Supplementary Table 11). Other measures included the DAWBA for psychiatric diagnosis and the TIMB interview for foster carer commitment. TTPLS and the child’s pre-care history of abuse and neglect were established by experienced social worker researchers scrutinizing each child’s social work casefile using the Adverse Childhood Experiences questionnaire and the Modified Maltreatment Classification System30. Data were collected at baseline (4–14 weeks after care entry), approximately 15 months after care entry and 2.5 years after randomization—face to face until March 2020 and then via telephone.
SAEs were recorded at research assessments, and, owing to concern about the level of risk in this population, a linkage to administrative data was made in November 2020.
Statistical analyses
The sample size was calculated by the Glasgow Clinical Trials Unit. Initially, the target sample size was 462 to achieve 90% power to detect an effect size of 0.35 s.d. on SDQ-TD with a loss to follow-up at 2.5 years of 25%26. With no information as to the likely degree of clustering of the primary outcome within families, we aimed to recruit 462 families, even though the unit of analysis would be the individual child. Although we later decided to constrain the sample size for financial reasons26, follow-up rates proved better than expected, and the trial was well powered. Given the s.d. of SDQ-TD observed in the trial at 2.5 years, a 0.35 s.d. difference equates to a clinically meaningful 2.5-point difference in the 20-item SDQ-TD scale.
A statistical analysis plan was prepared before database lock (see Supplementary Information). Clustering was at the level of the family because, if the family had more than one maltreated child, any effects of the NIM were likely to have an impact on every child. The primary analysis, conducted in R version 4.1.1 (2021-08-10) for Windows (with ‘lme4’ and ‘survival’ for TTPLS)31,32, used a generalized linear mixed effects regression model for the primary outcome measure (SDQ-TD), with random effects terms for both family and child, to account for clustering of outcomes within families and adjusting for child age and other minimization factors. All models were adjusted for age at measure completion. A single model over all three timepoints was fitted, and interaction terms were used to estimate intervention effects at each timepoint. Models were extended to include additional interaction terms to assess whether intervention effects varied between subgroups, for the following planned subgroup analyses for which sample size was sufficient: study site and sex. The primary analysis was intention-to-treat. To examine the causal effect of the NIM intervention in the presence of non-compliance, a CACE analysis was also conducted (using Applied Econometrics with R)33. This was achieved by comparing outcomes for individuals in the NIM group who complied with treatment (Supplementary Table 2) to individuals receiving SAU who were randomized to the NIM and would have complied with the NIM had they been given the opportunity to do so. A sensitivity analysis examined intervention effects for index children only. Bang’s unblinding index19 was used (using BI)19 to examine unblinding success.
We used the CONSORT framework to report the findings (see the checklist in Supplementary Information).
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
link